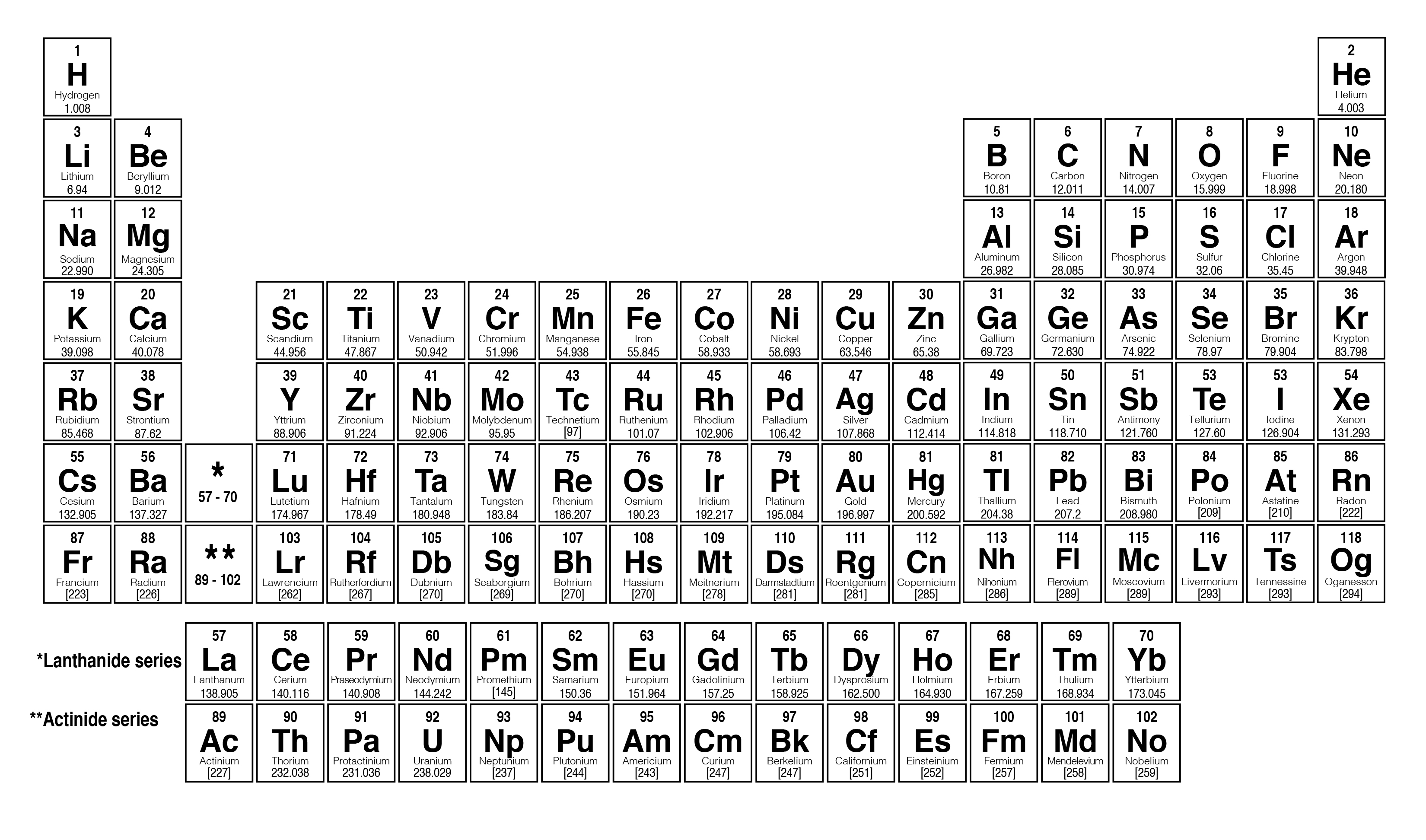
Again, the mass of a mole substance is called the molar mass, and to find the molar mass of a substance you just need a periodic table and the chemical formula. That’s the total molar mass of the compound CO2. Then finally you add all products, so 12 plus 32 is 44. The mole allows scientists to calculate the number of elementary entities (usually atoms or molecules ) in a certain mass of a given substance. You would multiply it by its subscript of 2, which is 32. Then you’d do the same thing with oxygen. Then the way you would find that is first you would find the atomic mass of carbon, which is 12, and then you’d multiply it by the subscript, which is just an understood 1 right there, so the product there would be 12. The molar mass has units of g/mol or kg/mol. If you were trying to find the molar mass of the compound carbon dioxide, which looks like this-CO2-and the molar mass is 44.
:max_bytes(150000):strip_icc()/PeriodicTableSigFigBW-58b5c7f25f9b586046cae098.png)
Then, to find the molar mass of the compound, you’re going to add all those products together. By multiplying the atomic mass by the subscript you’re getting the product, you’re getting the molar mass product for that atom. That way, you’re finding the total molar mass for all of that atom because you’re finding the atomic mass, then you’re multiplying it by however many times it appears in the compound. Generalizing this definition, the molar mass of any substance in grams per mole is numerically equal to the mass of that substance expressed in atomic mass units. After you’ve found the atomic mass, multiply the atomic mass by the subscript. You’re going to start with one atom at a time. I’m going to show you a 3-step process for finding the molar mass of a compound.įirst, find the atomic mass of an atom. You kind of follow the same protocol for finding the molar mass of an element, but then you combine all those molar masses together to find the molar mass of the compound. Now the molar mass of a compound can be calculated by summing the molar masses of each atom and the chemical formula. That’s how you find the molar mass of an element. When we add up the total values i.e, 46 + 12. 1 What is the molar mass of sodium carbonate, Na 2 CO 3 Solution Since sodium carbonate contains two atoms sodium, one atom of carbon and three atoms of oxygen. What is the formula to find molar mass The molar mass is calculated as the product of mass and moles. Add up all and assign unit as grams/mole. Now we’ll write it with a different unit because it’s also our molar mass, and that will be grams over moles: \(\frac\). The atomic masses can be found in the periodic table. When considering the element, aluminum, we can use the periodic table of elements to look up the atomic weight of this element (26.98 amu). If you’re looking at the element of copper you would look in the periodic table and find that it has an average atomic mass of 63.55 AMU, so that’s the molar mass. In such a conversion, we use the molar mass of a substance as a conversion factor to convert mole units into mass units (or, conversely, mass units into mole units). If you’re trying to find the molar mass of an element, then you’ll take the average atomic mass listed in the periodic table.
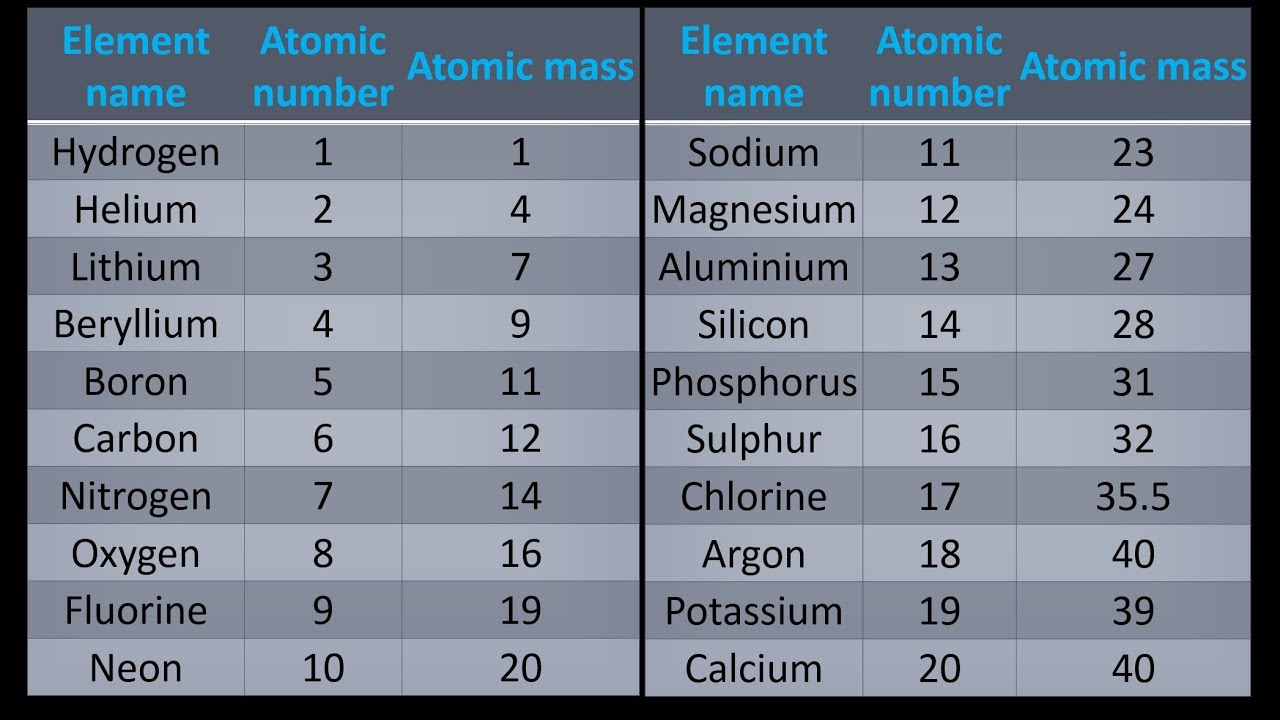
To calculate the molar mass of a substance you need a periodic table and the chemical formula. Element/Moleculeīy knowing the molar mass of a substance, having any mass of that substance allows one to know how many moles of it they have.The mass of a mole of substance is called the molar mass. (3 points per compound 12 points total) a. Using the periodic table calculate the molar mass of the following compounds. A list of some molar masses can be seen below. Youll get a detailed solution from a subject matter expert that helps you learn core concepts. The molar mass of any element or molecule is given by the sum of the atomic weights multiplied by 1 g/mol. In SI units, the molar mass is given by kg/ mol, however by convention the molar mass is expressed in units of g/mol. Now, compute the sum of products of number of atoms to the atomic weight: Molar mass (C 12 H 22 O 11 (sucrose)). Then, lookup atomic weights for each element in periodic table: C: 12.0107, H: 1.00794, O: 15.9994. This comparison is seen below:ġ dozen = 12 objects 1 mol = 6.022141 x 10 23 objects First, compute the number of each atom in C 12 H 22 O 11 (sucrose): C: 12, H: 22, O: 11. However when talking about moles, instead of only having a dozen, there is in fact 6.022141 x 10 23 of a given substance in a mole (known as Avogadro's number). For example, if someone has a dozen bricks and another person has a dozen feathers, they have the same amount of their substance, but a vastly different total mass. Since a mole is defined as the amount of a substance and substances have different masses, each element or molecule will have a different molar mass. Molar mass (M) is a physical property, defined as the mass of a given element or molecule per mole of that substance.
